Biology and movement of large brown trout in Lake Eildon

September 2008
John Douglas
Preferred way to cite this publication:
Douglas J (2008) Biology and movement of large brown trout in Lake Eildon. Fisheries Revenue Allocation Committee Final Report. (former) DPI, Victoria
ISSN 1833-3478
ISBN 978-1-74199-179-6
Table of Contents
Executive Summary
IntroductionObjectives
Methods
Trout population attributes
Growth
MortalitySpawning population age and length
Recruitment
Diet
Movement and habitat preference
Movement
Habitat preference
ResultsTrout population attributes
Growth
Mortality
Recruitment
Spawning population age and length
Diet
Movement and habitat preference
Discussion
Conclusions
Acknowledgements
References
Appendix 1
Appendix 2
Executive Summary
Lake Eildon in northeast Victoria supports a highly regarded brown trout (Salmo trutta) fishery. Recruitment of brown trout occurs annually from the tributaries, and the trout in the lake grow to reach ten years of age. Growth and mortality rates vary between years, and there is probably a relationship between lake water levels, productivity and summer water stratification.
The fishery for large brown trout is probably currently under-exploited. The larger fish are piscivorous and this diet may extend the growing season for the trout.
Lake stratification in summer can restrict the amount of habitat suitable for trout and prevent their access to the shallower parts of the lake. Trout are available to summer anglers if they understand how water temperature and lake stratification impacts on trout distribution.
Introduction
Lake Eildon in northeast Victoria is one of the State's premier recreational fisheries and attracts a large number of visitors to the area. There is a considerable amount of infrastructure in the area to support recreational fishing, and it is important that this fishery be maintained and enhanced both for the anglers and to support infrastructure and investment.
Lake Eildon is a significant and unique trout fishery, but it does produce some excellent large brown trout (Salmo trutta) but does not produce good quality small fish. Summer surface water temperatures are high and the large fish can be hard to find at this time.
The aim of this study was to answer the following research questions.
- What is the biological basis for the observed population structure and can anglers sustainably harvest more large brown trout in the lake?
- Where are these large brown trout located and what are they feeding on; once known, anglers can be advised on where and how to target them more effectively?
Objectives
The main objective of this project was to directly benefit anglers by determining where large brown trout prefer to be and what they feed on. This will assist anglers to efficiently target these larger fish.
To attain this main objective, the following objectives were formed:
- To describe biological attributes of the brown trout population in Lake Eildon.
- To describe brown trout recruitment patterns.
- To describe feeding preferences of large brown trout in the lake.
- To investigate habitat preferences of large brown trout.
- To provide advice that will assist anglers to catch more large brown trout.
Methods
The overall design of the study centred on:
- obtaining length-at-age information for the Lake Eildon brown trout population, investigating growth rates, mortality rates and identifying successful year-classes
- investigating the age structure of the spawning brown trout population to determine the significance of large trout as broodstock
- monitoring the movement of large brown trout within the lake to gain insights into the areas and habitats that they frequent
- undertaking diet studies to investigate any differences in the diets of different sized fish
- determining the suitable habitat of trout.
Trout population attributes
Growth
Trout ageing was undertaken using otoliths. Trout otoliths, collected from previous studies (1999) in Lake Eildon were collated. Age was estimated from thin-sections by the Central Ageing Facility (CAF), Queenscliff. Von Bertalanffy growth equation length-at-age was produced for the dataset.
Mortality
Instantaneous mortality rates are used to describe the decrease in numbers of fish over time, or to calculate the number of the fish remaining at the end of a period of time. The instantaneous total mortality rate is composed of natural mortality rate (fish that die of natural causes) and the mortality rate caused by fishing (fish taken by anglers). An estimate of the instantaneous total mortality rate (Z year-1) was calculated for Lake Eildon, by conducting a regression on the age-class frequencies from the aged trout samples. The negative slope of the curve is the instantaneous mortality rate (Z) (Ricker 1975).
Longevity is an estimate of length of life and was calculated from an empirical equation (Gayanilo and Pauly 1997): tmax≈3/K where tmax is the approximate longevity, and K the rate at which the fish reaches its asymptotic mean length at the theoretical age of infinity.
Spawning population age and length
Age and size structure of the present spawning population in Lake Eildon was determined by collecting a sample of trout from the spawning run in June 2004. Samples were collected by boat mounted electrofishing at the mouths of the Goulburn and Howqua Rivers. In June 2004, a sample of trout was also obtained by back-pack electrofishing from Still Creek, a tributary of the Big River that flows into Lake Eildon. A further sample of trout from Still Creek was obtained in February 2005 and an ANOVA was used to investigate any differences in the mean ages between these trout and to confirm that the larger fish observed in winter were not resident fish in the stream, and therefore likely to be spawning fish migrating up the tributary streams from Lake Eildon.
Recruitment
Age-frequency analysis was used to investigate the better recruitment years. Trout otoliths collected from the spawning run during the present study were aged at the CAF. Lake height was compared with the age frequency to investigate any coarse relationship between lake level and better recruitment years.
Diet
Monthly netting surveys of Lake Eildon were undertaken from May 2004 to April 2005. A fleet of nets was set perpendicular to the shore and checked daily for two days. All trout captured were weighed and measured and the stomach contents assessed. Food items that could be identified in the field were identified and ranked in order of volume. If a positive identification could not be made the prey item was classified as unidentified. Food items were classified according to their source as benthic, pelagic, terrestrial, fish and other as per Douglas et al (2002).
Movement and habitat preference
Movement
Ten large trout (> 2.5 kg) were collected by mesh netting or boat-based electrofishing. Upon capture, trout were anaesthetised with Benzocaine (ethyl p-aminobenzoate) at concentration of 0.05–0.1 g per litre of water. An acoustic transmitter (pinger) was implanted surgically through a 25 mm incision on the ventral side of the fish. The pinger was inserted entirely into the abdominal cavity and the incision was closed by sutures and then further sealed with a cyanoacrylate based adhesive. Total surgery time averaged two to three minutes. The fish were kept moist with constant spraying of water, via an atomiser, over the gills and Biology and movement of large brown trout in Lake Eildon body while the surgery was in progress. After surgery, anaesthetised fish were allowed to recover in an aerated fish bin. Fish were released at their capture site. Fish were not examined to determine sex, therefore no differentiation is made between sexes. The transmitters could be detected with a hand held uni-directional hydrophone and had a range of several hundred metres. Once a fish was located, a directional hydrophone was employed and the location of the tag could be determined to within 20 metres. The transmitters were coded so individual fish could be identified.
Fish were monitored on a monthly basis by driving a boat around the lake and listening for fish signals at regular intervals with the unidirectional hydrophone. If a fish was detected, the location was recorded with a handheld global positioning system unit (GPS) and the lake depth read from the boat's depth sounder.
Habitat preference
Suitable trout habitat was estimated by determining the vertical extent of suitable temperature and oxygen levels based on measured water temperature and dissolved oxygen profiles obtained from the lake. The area and location of this segment of "suitable habitat" was then calculated by applying the lake level and bathymetry of the lake bed information using geographical information system software (Arcview 3.2). Water temperature profiles, lake level and bathymetry data were obtained from Goulburn-Murray Water. A water temperature of 19 0C was chosen as the upper preferred water temperature limit for the lake brown trout as this figure is considered to be at the top end of optimum growth and where feeding ceases (Elliott and Hurley 2000). The minimum dissolved oxygen level for trout was chosen to be 3 ppm (Raleigh et al 1984 in Dedual et al 2000). Dissolved oxygen profiles were not available for Lake Eildon. Dissolved oxygen is often depleted in the hypolimnion below the thermocline (Wetzel 1983) and an estimate of the depth of the hypolimnion was made after a visual assessment of the Lake Eildon temperature profiles for both summer and winter. For this study, the depth of the top edge of the hypolimnion was at a depth of 30 metres in winter and at a depth of 25 metres in summer. These figures were derived from Lake Eildon water and oxygen profiles supplied by Goulburn Murray Water.
Suitable habitat for trout was considered to be the strata that were bounded between the top edge of the hypolimnion and the 19 0C isotherm. Using GIS, the habitat strata information was modelled with the lake bathymetry to delineate and diagrammatically render the amount and location of suitable brown trout habitat for the whole lake. This method was applied to winter and summer water temperature profiles to quantify any changes in available habitat on a seasonal basis. For the purposes of this study, summer was when the lake was highly stratified from December to March; winter was from April to November.
Brown trout are pelagic but make greater use of the littoral zone than rainbow trout (Graynoth 1999). The benthic area available to trout, i.e. the area of the lake-bed that was within the suitable habitat strata, was calculated for summer and winter. This habitat reduces by a similar magnitude to the overall habitat (Table 2) when the lake stratifies.
Results
Trout population attributes
Growth
The Lake Eildon trout population is made up of a range of fish ages and lengths (Figure 1 and 2) and growth rate varies across time. Brown trout are generally less than 600 mm in length. Some fish that grow beyond this mark. The most frequent age in the population was three year old fish. This compares with the historic aged brown trout data (Figure 3). Growth rates for the 2005 fish were higher than the historic sample with three-year-old fish reaching nearly 500 mm (range 250–480 mm) (Figure 2) compared with the historic three-year-old brown trout that failed to reach 400 mm (range 210–390mm) (Figure 4). The oldest fish aged from Lake Eildon was 10 years. Brown trout older than seven years were not recorded in the current sample. Lake Eildon trout can reach up to 600 mm in length, and this has remained similar between the historic and recent data (Figure 5 and Figure 6).
About half the fish were younger than three years and for the other half of the population, the ages ranged from four to seven years of age. Longevity was calculated with Pauly's M equation and was estimated at 10 years. This is also the age of the oldest fish aged from the historical data (Figure 2).
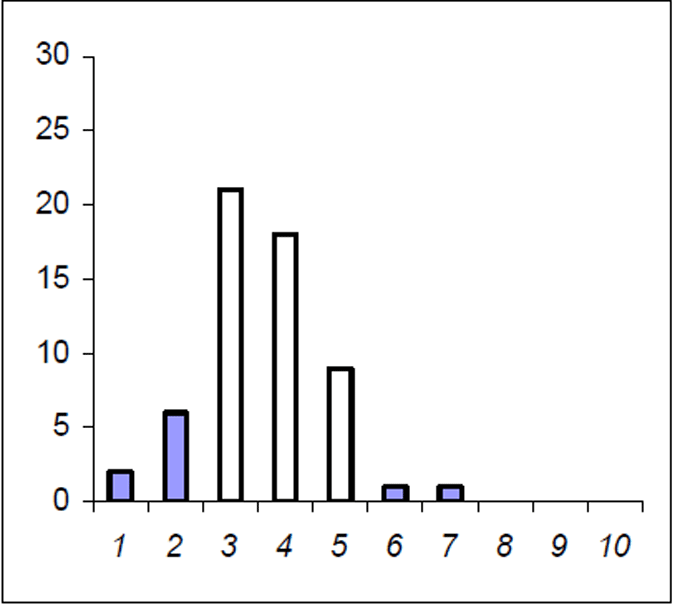
Figure 1. Age frequency for Lake Eildon brown trout in 2005. Frequency on Y axis
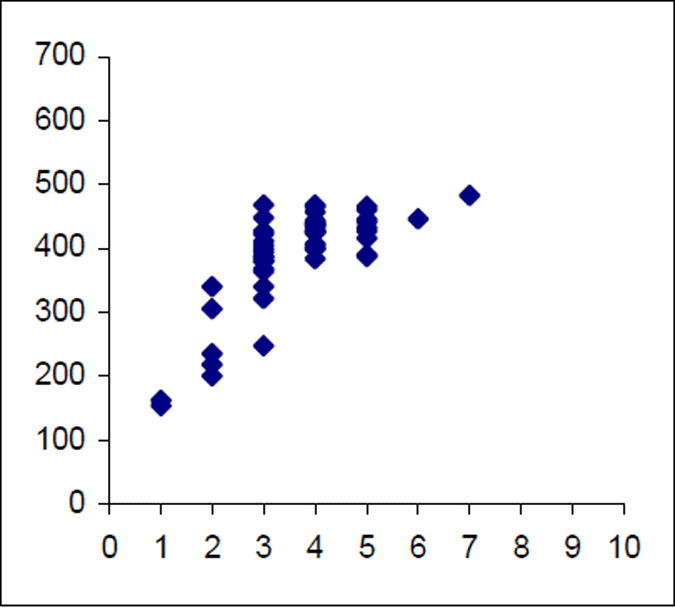
Figure 2. Length–age scattergram for Lake Eildon brown trout in 2005. Age on X axis
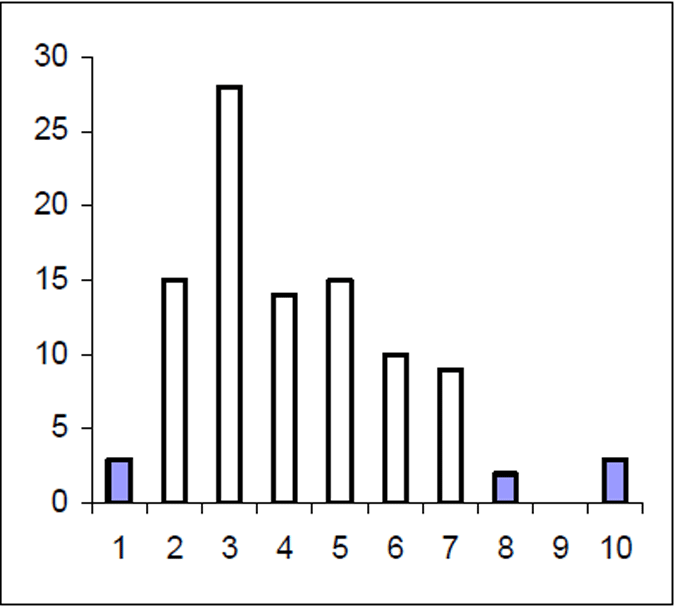
Figure 3. Age frequency for Lake Eildon brown trout in 1999. Frequency on Y axis
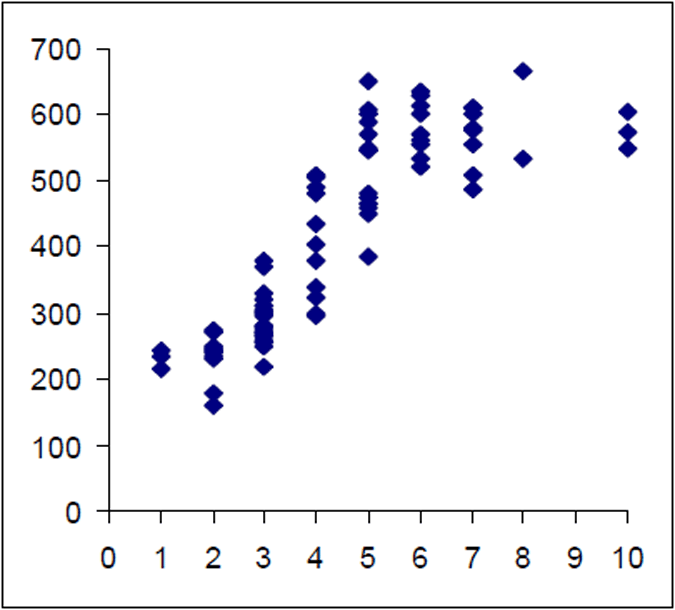
Figure 4. Length–age scattergram for Lake Eildon brown trout in 1999. Age on X axis.
The von Bertalanffy growth parameters for Eildon brown trout were calculated as:
L∞ =644.3 (mm), K=0.3005 year-1 and T0 =0.4707 year
There is disparity in growth rates, and length-atage results indicate brown trout in Lake Eildon have variable growth. Brown trout in Lake Eildon can reach a length of 500 mm (lcf) in their sixth to seventh year. Age-frequency distribution shows a decline in abundance with age and no fish were found to be older than 7 in the current survey.
Mortality
The mortality rate of the historical (1999) Lake Eildon brown trout sample was calculated as Z=0.416. That is equivalent to an annual mortality rate of 34% for fish between 3 and 10 years of age (Figure 7). The instantaneous mortality rate of the 2004–05 sample, based on estimated ages from an age length key, was calculated as Z=0.763 that is equivalent to an annual mortality rate of 53% for brown trout aged between four and 10 years of age (Figure 8). Brown trout mortality rates varied over time in Lake Eildon.
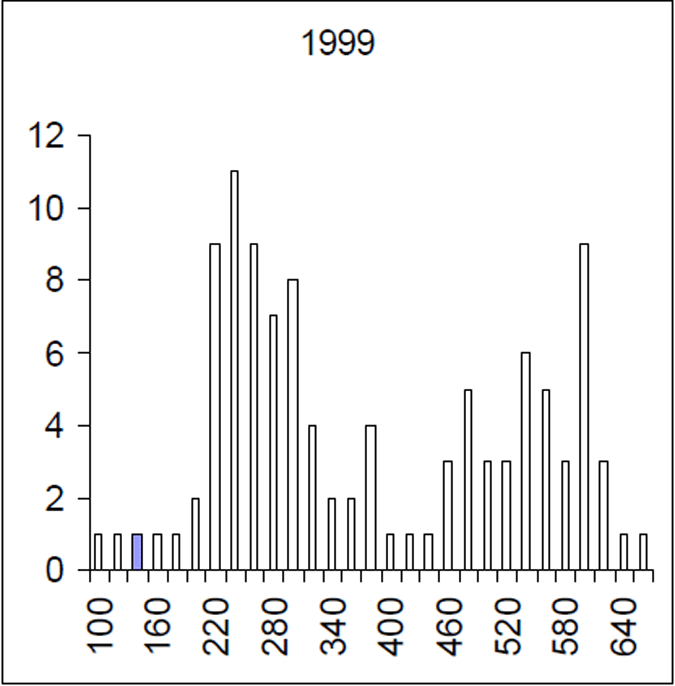 Figure 5. Length frequency of 1999 brown trout sample. Length is lcf in millimetres. (n= 228)
Figure 5. Length frequency of 1999 brown trout sample. Length is lcf in millimetres. (n= 228)
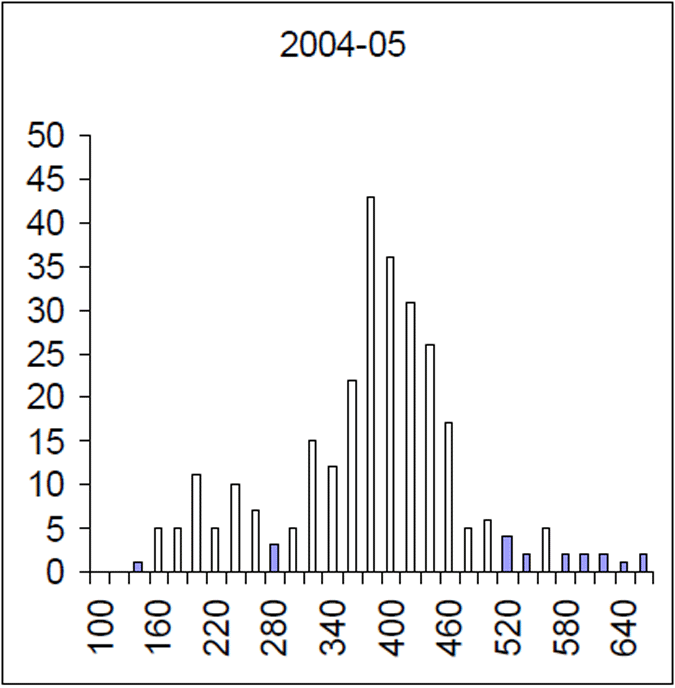
Figure 6. Length frequency of 2004–05 brown trout sample. Length is lcf in millimetres. (n=256)
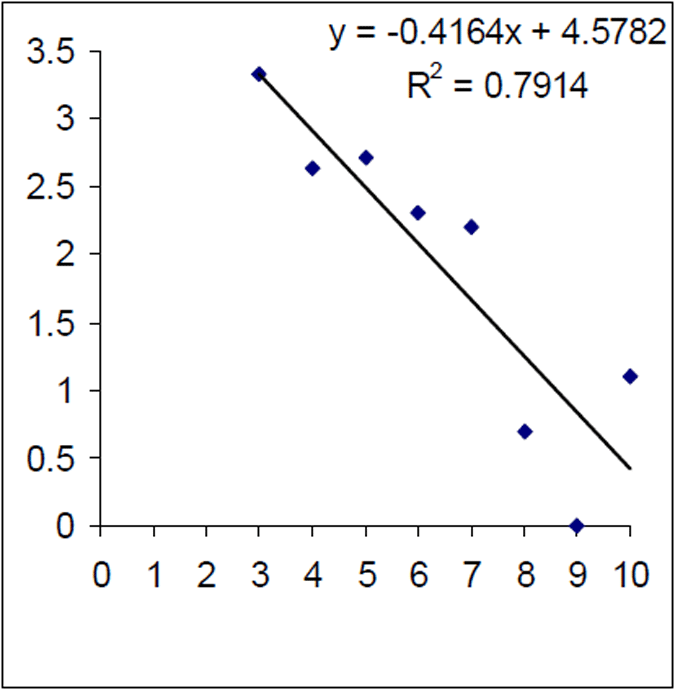
Figure 7. Log of age frequency and regression from historical (1999–2000) Lake Eildon brown trout sample (n=99). X axis is age in years
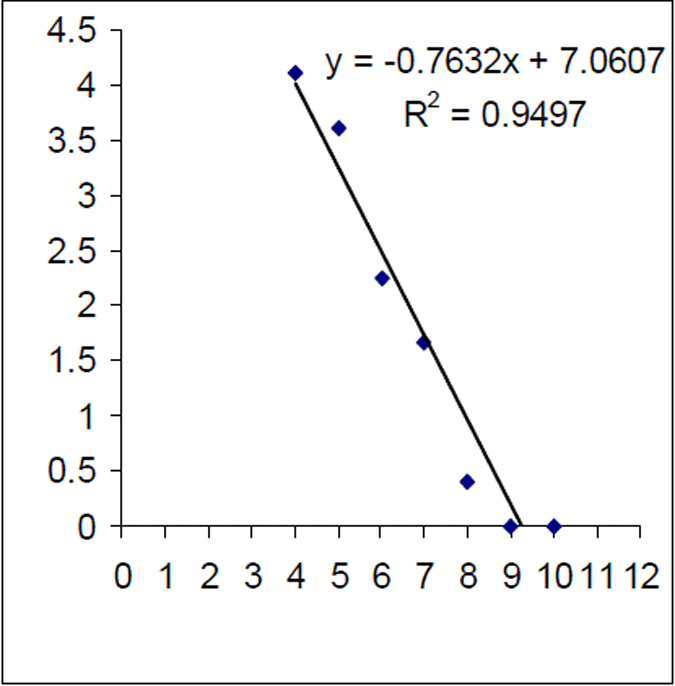
Figure 8. Log of age frequency and regression from 2004–05 Lake Eildon brown trout sample (n=256) (age estimate based on age length key). X axis is age in years
Recruitment
Spawning population age and length
Recruitment of brown trout in Lake Eildon is usually both annual and successful in most years, as evidenced by the presence of fish from each birth year back to 1989 excluding 1990. There were insufficient data to investigate annual recruitment with lake heights as a time series of trout samples was not available.
Relatively young fish dominate the sample of the trout spawning run of Lake Eildon trout (Figure 9). The ages of these spawning run fish are generally under five years of age, with the highest age frequencies in the two and three year old categories (e.g. maiden fish). Although present in the lake, older fish do not appear to represent a large proportion of the spawning fish population.
The ages of trout in Still Creek vary between winter and summer (p=0.01). The mean age of the winter sampled brown trout in Still Creek are not different from the mean ages of spawning run brown trout sampled at the mouths of the Howqua and Goulburn Rivers (p=0.29). The fish in Still Creek in winter are likely to be spawning trout from the lake and are itinerant, whereas the summer sample is likely to represent resident creek fish.
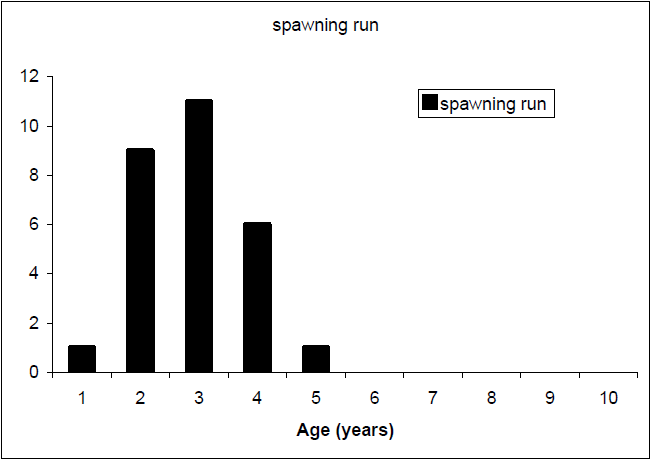
Figure 9 Age frequency of spawning run brown trout sample (n=28),
Diet
The diet of Lake Eildon trout is principally fish (Figure 10). Terrestrial and pelagic items are present but were proportionally low relative to fish food items. Of the fish in the diet, most items were well digested and could not be easily identified. Of the fish identified, redfin were the most common prey species. Roach and goldfish were identified but in low proportions compared with the overall diet (3%). Pelagic items, which included aquatic invertebrates such as cladocerans, were the least recorded food category.
The diet of Lake Eildon trout varied slightly over the year with more terrestrial food consumed in the winter months (April–November) (Figure 11). Fish is the dominant food source for trout in Lake Eildon over the summer (December–March). Fish was the dominant component of the diet of brown trout of length more than 500 mm (based on a limited sample, n=13).
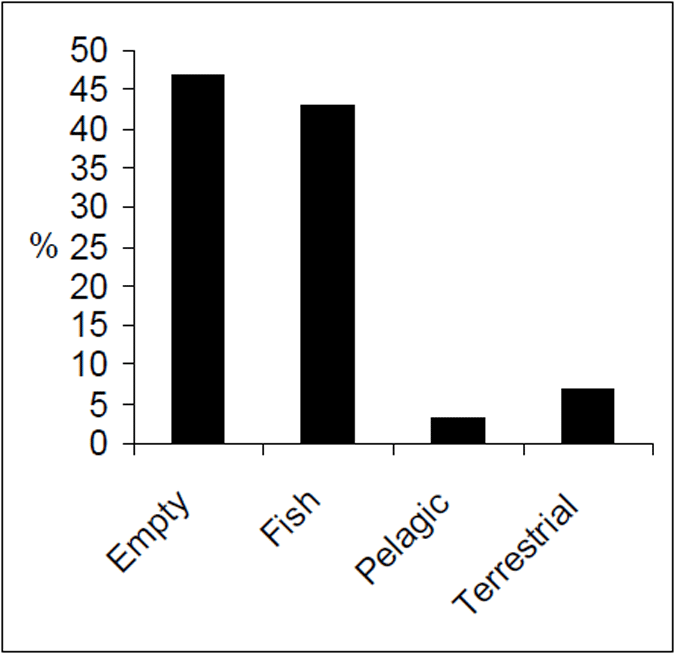
Figure 10. Food categories by percent for brown trout from Lake Eildon (n=128).
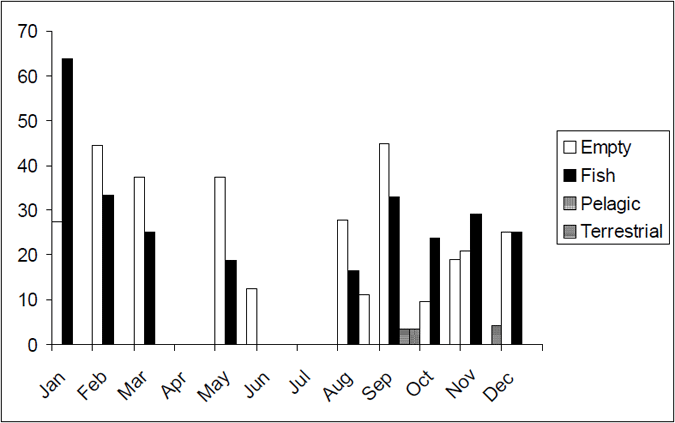
Figure 11. Seasonal food categories by percentage for brown trout from Lake Eildon (n=128)
Movement and habitat preference
Ten trout were fitted with acoustic tags and released. Two fish were not located again and although eight fish were subsequently tracked, a further four fish either died over the course of the tracking program did not contribute long term movement data. Tracking maps for the eight fish monitored are presented in Appendix 1. Four fish were tracked for extended times and summaries of these fish are presented in Table 1. Of these tracked fish, the most number of times a fish was located was 24 (BT16) and the least number of times fish were located was 12 (BT12 and BT18).
The brown trout did not display any home range behaviour and were generally found to roam over large areas of the lake. The fish that were tracked the most (BT 16 and BT 17), used the entire length of the lake including some of the inflowing tributaries. There was no obvious association or affinity of trout locations with any particular lake feature or area. The trout generally moved around and did not stay in any area for long. In summer the range of trout appeared to be confined to the main body of the lake because no trout were located in the shallow lake inlets.
The trout movement data were compared with a prediction of suitable habitat area for brown trout that was modelled for Lake Eildon. The habitat model was based on lake bathymetry, lake height, and summer and winter water-temperature profiles from Lake Eildon. Applying the trout temperature preferendum to the temperature related depth constraints indicated that in winter, trout habitat extended from the water surface down to about 30 metres. In summer the increased temperature of the warmer surface layers push the 190C isotherm to 15 metres deep and compress suitable conditions for trout to a 10 metre strata between this isotherm and the top of the hypolimnion at 25 metres deep.
GIS analysis of these strata and the underlying bathymetry indicated that suitable trout habitat changed considerably between summer and winter. The predicted summer and winter trout habitat area is presented in Table 2. In summer, the area of suitable trout habitat was reduced by 71% from the winter habitat and was restricted to the main body of the lake and excluded the shallow inlets.
The benthic area available to trout, i.e. the area of the lake-bed that was within the suitable habitat strata, was also calculated for summer and winter. This habitat reduces by a similar magnitude to the overall habitat (Table 2) when the lake stratified.
Six of the eight tagged brown trout provide suitable data to be used in the analysis. Comparing the summer and winter locations of these six trout, there is a decrease in range between the seasons. When the summer locations of brown trout were compared with the predicted habitat, there was a very high correlation between the locations and the predicted habitat. No trout was found outside the predicted summer habitat range.
Table 1. Summary of acoustic tagged trout tracking data.
| Trout No | Release date | Last date located |
Number of times fish located |
|---|---|---|---|
| BT16 | 14/8/02 | 23/4/03 | 24 |
| BT17 | 14/8/02 | 3/3/03 | 20 |
| BT20 | 14/8/02 | 18/3/03 | 17 |
| BT21 | 20/9/02 | 3/3/03 | 17 |
Table 2. Estimated reduction in area of predicted winter and summer brown trout habitat in Lake Eildon 2004.
| Habitat | Winter Area (Ha) | Summer Area (Ha) | Reduction in area (%) |
|---|---|---|---|
| All | 5103 | 1471 | 71 |
| Benthic only | 3632 | 885 | 76 |
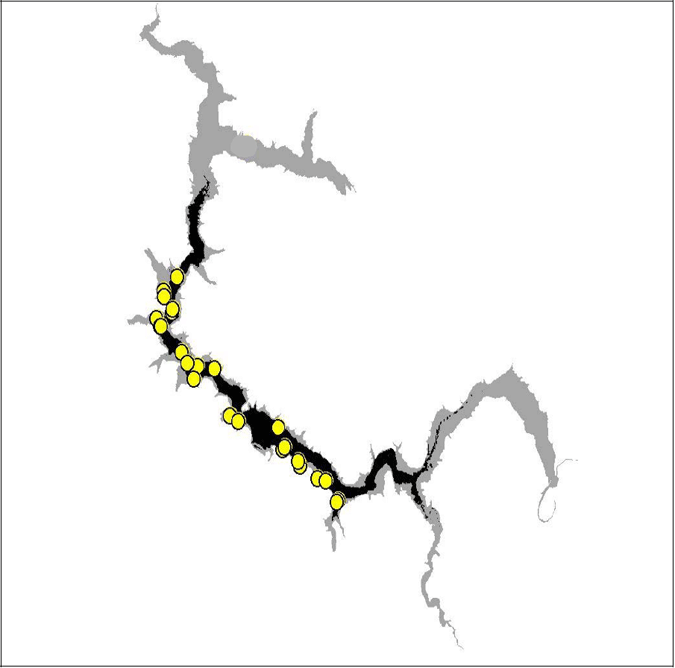
Figure 12. Diagram of predicted summer habitat (black) and observed summer trout locations in Lake Eildon 2003–04
Discussion
Growth
The growth model for brown trout in Lake Eildon indicates that they have the potential to attain a predicted asymptotic average length of just over 600 mm. Fish of this size are highly sought by trout anglers. The longevity of these Lake Eildon trout is up to 10 years of age and this is likely to reflect the lake dwelling habit of the fish and on a world scale is normal. Brown trout may reach ages up to 20 years but most die before this and few fish live longer than about 10 years (Klemetsen 2003). Compared with stream trout, lake living trout are usually longer lived and mature later (Klemetsen 2003).
Intra-year class competition in juvenile salmonids impacts on size classes as larger juveniles commonly out-compete smaller cohort members and leads to high mortality in the slow-growing subordinate fish (Reinhardt et al 2001) and this may have been a factor as the brown trout population structure in 1999 contained many more year-classes than when sampled in 2004–5. Reduced competition among fewer year-classes for the available food resources may partly explain the faster growth rates seen in the recent samples.
Diet
The diet of large trout in Lake Eildon is predominantly fish, and a fish diet allows trout to utilise slightly warmer temperatures than a nonpiscivorous diet. A piscivorous diet may extend both the growing season and the area of suitable habitat available for trout in Lake Eildon. By being piscivorous, rather than feeding on invertebrates, trout greatly increase their energy intake and can eat less food to receive the same energy (Elliott and Hurley 2000). Warm-water temperatures are known to suppress feeding in many species, including trout (McMichael and Kaya 1991). A diet of fish increases the optimum temperature for growth of trout (Elliott and Hurley 2000) and therefore, may be beneficial for growth in Lake Eildon trout by allowing feeding at higher temperatures. The morphology of the reservoir and the summer draw-down are not suitable for the formation of extensive weed beds and this is likely to suppress macroinvertebrate production. Invertebrates recorded from the diet indicate that terrestrial insects are an important diet supplement and aquatic insects appear to be relatively rare. Daphnia were not prevalent in the diet of Lake Eildon trout. This contrasts to the diet of trout in Lake Dartmouth in north east Victoria where cladocerans and other pelagic foods were significant items in the diet (Douglas et al 2002). Energy intake, growth, and the optimum temperature for growth increase markedly when trout change their diet from invertebrates to fish (Elliott and Hurley 2000). The lack of invertebrate food is not favourable to small trout and those that switch to a fish diet early may benefit in terms of growth and their temperature tolerance. The paucity of invertebrates in the diet may be the reason that the lake does not support a good small trout fishery.
Exploitation
In a comparative study of exploited and unfished trout populations, exploited populations showed a significant decrease in age structure diversity, life span, and the proportion of individuals at length above the legal minimum length in contrast to the unexploited population (Braña et al 1992). The population structure of the Lake Eildon trout indicates that the fishery is not currently over exploited because of the age diversity and presence of large fish.
Spawning and recruitment
The large trout make only a small proportion of the spawning run, so it is likely that the recreational take of these fish could be increased without significantly impacting on recruitment levels. There is some evidence that larger fish produce bigger eggs (Pittman 1979) with subsequent lower mortalities in the life cycle stage between fertilisation and first feeding providing a competitive advantage post-hatching (Elliott 1984). The increased fecundity of the larger fish and the increased survival of their offspring could, therefore, significantly contribute to overall recruitment. Large eggs and subsequent large fry can provide a recruitment advantage (Elliott 1984). On the other hand, trout recruitment may not be density dependent and removal of some of the spawning population may not decrease recruitment levels. An example of this was provided in Klemetsen et al 2003, who reported that population recruitment of brown trout remained the same even when a large proportion of the spawners were removed.
In lakes such as Lake Eildon, the wild fishery depends on juvenile trout moving from the natal streams into the lake. In many lake systems, large numbers of fry migrate to the lake immediately after emergence; however, these fish may only make up a small proportion of recruits (Hayes 1995). Early life stages in fish make an important contribution to production, but mortality is usually great during the early stages of life and variation in early-life-stage mortalities can lead to year-class fluctuations (Bagenal and Braum 1978). Strategies that minimise early life stage mortalities can increase year-class strengths and some trout employ such approaches. In some fisheries, brown trout stay in the tributary streams for one to two years before recruiting to the lake (Graynoth 1999, Douglas et al 2002). A period of stream residence, allowing juveniles to grow to a particular size threshold, was found to assist in trout survival in some lakes (Northcote 1978, Rosenau 1991). Recruiting to the lake at a larger size has survival advantages, particularly if there is a lack of invertebrate food for young or small fish. The larger fish can switch to a fish diet as soon as they enter the lake. In comparison, stream dwelling has some disadvantages in shortened longevity and slower growth (Jensen and Johnsen 1999). Reduction in the proportion of energy available for growth has been proposed as an explanation that initiated trout to move out of streams (Forseth et al 1999).
Catchment conditions suitable for prolonged stream residence (e.g. suitable temperatures, flows and habitat), prior to lake recruitment may ultimately provide for a diverse age-structure (and more big fish) within the lake population. However, this multi-age-class structure may suppress individual growth rates through intra year-class competition as discussed above. Recent prolonged drought conditions within the Lake Eildon catchment have probably been unsuitable for prolonged stream residence of young fish, and may explain the difference in population structure and as seen between 1999 and 2004–05 samples. Elliott et al. (1997) suggested that 0+ and 1+ stages of brown trout were negatively affected by summer droughts—possibly by a reduction of available suitable natal stream habitat.
Irrespective of how long the fish reside in these streams, the importance of the inflowing tributary streams for the trout recruitment in Lake Eildon is clear. Processes that degrade these areas will impact on recruitment and thus impact on the sustainability of the fishery. Therefore, any stream habitat or catchment activities have the potential to impact on the fishery, and any actions or activities that improve the habitat condition in these streams would most likely the fishery.
Movement
Acoustic tagging of brown trout indicated that the trout use much of the available lake but do not remain in the extremities of the inlets in summer. In winter the trout are widely distributed throughout the lake including the inlets, but were only detected in the main lake in summer and not in the inlets. The acoustic tracking also indicated that trout did not display behaviour that could be interpreted as site affinity. It was concluded that they did not have home ranges, but were nomadic and covered large areas. The tagged trout were often located near the shore (within approximately 200 metres) and this agreed with the findings of Dedual et al (2000) who observed New Zealand brown trout to be more sedentary than rainbow trout and make more use of the littoral zone. They reported home range and homing abilities in brown trout, but no such behaviour was observed during this study. They also speculated that the trout distributions were related to food resources and the fish distributions correlated with areas where there was an abundance of preferred prey, or prey of a large size. Lake Eildon trout are piscivorous, whereas the New Zealand trout were invertebrate feeders. Lake Eildon does not have the extensive weed beds of New Zealand lakes and macroinvertebrate production may be relatively low as a result; this is supported by the low frequency of benthic organisms in the Lake Eildon trout. A piscivorous diet may force trout to follow the food source and limit the development of home ranges or territories.
Trout in Lake Eildon were predominantly piscivorous and their distribution is likely to be linked to the distribution of prey (food supply), which includes redfin and roach. In Lake Eildon, schools of small redfin (up to about 12 cm in length) are often observed near the shoreline or in the branches of sunken trees. Dietary observations indicated redfin as a major food source suggesting the distribution of redfin would influence the distribution of trout. Little is known of redfin movements in Lake Eildon. Cadwallader and Backhouse (1983) suggested that redfin are often associated with cover, although they may also be pelagic in lakes. They also reported that adult redfin may move into shallow water in spring, and possibly stay in such habitat for some time. Juvenile redfin have been found to aggregate in dense schools in the warmer parts of the water during the day in European lakes (Masson 2001). Small roach are planktivores and tend to be associated with concentrations of plankton, although diet can change with size and larger roach tend to eat more benthic organisms such as snails (Pers comm Daniel Stoessel PIRVic – Snobs Creek).
Habitat
Previous habitat preference investigations suggest that oxygen and temperature influence the available lake habitat to trout (Rowe and Chisnall 1995). In large temperate impoundments such as Lake Eildon, stratification can lead to a 'squeeze' in habitat in summer, as the surface waters are too warm and the oxygen levels in the lower water are depleted. Impacts from this temperature– oxygen 'squeeze' include suppressed feeding (Headrick and Carline 1993) and even possible death from malnutrition (Zale et al 1990). The summer habitat 'squeeze' would be expected to impact growth and survival of trout in Lake Eildon in most summers. The severity of the summer habitat restriction in Lake Eildon would be linked to the environmental conditions of the summer; i.e. the degree to which water temperatures were above or below average temperatures, and the concurrent lake water levels. This impact would vary with environmental conditions and the effect on the Lake Eildon trout populations would also vary.
Trout anglers should be aware of trout temperature preferences to improve their chances of locating trout (Jones 1982). Apart from a reduction in habitat size, the restriction in habitat also restricts the distribution of trout. In Lake Eildon, trout anglers should be aware of the summer habitat restriction and confine their angling efforts to where the trout are located. In the summer months, they should concentrate their efforts in the main body of the lake and stay out of the shallow inlets.
Generally, trout anglers do not fish in the lake during summer. A key aim of this project was to assist anglers to target the larger trout more effectively. Current angling methods are probably satisfactory for winter; however, the fish are still available in summer. Anglers wishing to target trout in Lake Eildon in summer, should endeavour to locate concentrations of prey such as redfin within the summer habitat range of the trout. This translates to concentrating effort in the strata above the thermocline and below about 190C water temperature (~15 m).
Conclusions
The Eildon brown trout fishery is dependant upon recruitment from the tributary streams and the condition of these streams is very important to the fishery.
- The brown trout population is not currently over harvested.
- Large brown trout in Lake Eildon are generally piscivorous, and redfin is a large component of their diet.
- A piscivorous diet may assist brown trout to grow larger in temperate impoundments such as Lake Eildon.
- Lake Eildon brown trout roam around the available habitat, and do not display any home range behaviour.
- The available habitat to trout is restricted in summer due to lake stratification and surface layer water temperature.
- Large brown trout are available to recreational anglers all year, but in summer anglers should utilise methods that can concentrate their efforts in the deeper areas of the lake located above the thermocline and below the 19°C water temperature.
Acknowledgements
Thanks are given to Russell Strongman, Karl Pomorin, Daniel Stoessel and Andrew Pickworth who undertook the surgery, fish tracking and trout sampling work. Karl Pomorin and Nigel Abery conducted the GIS analysis. Paul Brown, Wayne Fulton and Kylie Hall provided comments and suggestions on the manuscript and provided editorial contributions.
This project was funded by revenue raised from the sale of Victorian Recreational Fishing Licences.
References
Bagenal TB and Braum E (1978) Eggs and early life history. In 'Methods for assessment of fish production in freshwaters' (T Bagenal Ed.), 3rd edition, pp165–201. IBP Handbook No 3, Blackwell Scientific, Oxford.
Braña F, Nicieza AG and Toledo MM (1992) Effects of angling on population structure of brown trout, Salmo trutta L., in mountain streams of Northern Spain. Hydrobiologia 237, 61–66
Cadwallader PL and Backhouse GN (1983) 'A guide to the freshwater fish of Victoria'. (Ministry for Conservation, Victoria). 249pp.
Dedual M, Maxwell ID, Hayes JW and Strickland RR (2000) Distribution and movements of brown trout (Salmo trutta) and rainbow trout (Oncorhynchus mykiss) in Lake Otamangakau, central North Island, New Zealand. New Zealand Journal of Marine and Freshwater Research 34, 615–627
Douglas J, Giles A and Strongman R (2002) Lake Dartmouth multi-species fishery assessment. Marine and Freshwater Research Freshwater Fisheries Report 02/2.
Elliott JM (1981) Some aspects of the thermal stress on freshwater teleosts. In AD Pickering (Ed) Stress and Fish. Academic Press, London. Pp 209–245
Elliott JM (1984) Numerical changes and population regulation in young migratory trout Salmo trutta in a Lake district stream, 1966-83. Journal of Animal Ecology 53, 327–350
Elliott JM and Hurley MA (2000) Daily energy intake and growth of piscivorous brown trout, Salmo trutta Freshwater Biology 44, 237–245
Elliott JM, Hurley MA and Elliott JA (1997) Variable effects of droughts on the density of a sea-trout Salmo trutta population over 30 years. The Journal of Applied Ecology 34, 1229–1238
Forseth T, Naesje TF, Jonsson B, and Harsaker K (1999) Juvenile Migration in Brown Trout: A Consequence of Energetic State. The Journal of Animal Ecology 68, 783–793
Gayanilo FC Jr and Pauly D (1997) FAOICLARM stock assessment tools (FISAT). Reference manual. FAO Computerised Information Series: No 8. Rome, FAO 262p.
Graynoth E (1999) Recruitment and distribution of juvenile salmonids in Lake Coleridge, New Zealand. New Zealand Journal of Marine and Freshwater Research 33, 205–219 Hayes J (1995) Importance of stream versus early lake rearing for rainbow trout fry in Lake Alexandrina, South Island, New Zealand, determined from otolith daily growth patterns.
New Zealand Journal of Marine and Freshwater Research 29, 409–420
Jensen AJ and Johnsen BO (1999) The functional relationship between peak spring floods and survival and growth of juvenile Atlantic Salmon (Salmo salar) and Brown Trout (Salmo trutta). Functional Ecology 13, 778–785
Jones AR (1982) The "two-story" rainbow trout fishery at Laurel River Lake, Kentucky. North American Journal of Fisheries Management 2, 132–137
Klemetsen A, Amundsen PA, Dampson JB, Jonnson B, Jonsson N, O'Connell MF and Mortesen E (2003) Atlantic salmon Salmo solar L., brown trout Salmo trutta L. and Artic charr Salvelinus alpinus (L.): a review of aspects of their life histories. Ecology of Freshwater Fish 12, 1–59
McMichael GA and Kaya CM(1991) Relations among stream temperature, angling success for rainbow trout and brown trout, and fisherman satisfaction.North American Journal of Fisheries Management 11, 190–199
Masson S, Angeli N, Guillard J and Pinel-Alloul B (2001) Diel vertical and horizontal distribution of crustacean zooplankton and young of the year fish in a sub-alpine lake: an approach based on high frequency sampling. Journal of Plankton Research. 23(10) 1041
Northcote TG (1978) Migration strategies and production in freshwater fishes. In: Gerking. S.D. ed., Ecology of Freshwater Fish Production, pp. 326–359. Oxford, Blackwell Scientific Publications.
Pittman RW (1979) Effects of female age and egg size on growth and mortality in rainbow trout. The progressive fish culturist 41, 202–204
Ricker WE (1975) Computation and interpretation of biological statistics of fish populkations. Bulletin of the Fisheries Board of Canada 191, 382pp.
Rosenau ML (1991) Natal-stream rearing in three populations of rainbow trout in Lake Taupo, New Zealand.New Zealand Journal of Marine and Freshwater Research 25, 81–91
Rowe DK and Chisnall BL (1995) Effects of oxygen, temperature and light gradients on the vertical distribution of rainbow trout,
Oncorhynchus mykiss, in two North Island, New Zealand, lakes differing in trophic status. New Zealand Journal of Marine and Freshwater Research 29, 421–434
Reinhardt UG, Yamamoto T, Nakano S (2001) Effects of body size and predators on intracohort competition in wild and domesticated juvenile salmon in a stream.Ecological Research 16 (2), 327–334 Wetzel (1983) Limnology. 2nd Edition, Saunders College Publishing.767pp.
Zale AV, Wiechman JD, Lochmiller RL and Burroughs J (1990) Limnological conditions associated with summer mortality of striped bass in Keystone Reservoir, Oklahoma. Transactions of the American Fisheries Society 119, 72–76
Appendix 1
Locations of individual trout tracked in Lake Eildon between August 2003 and March 2004
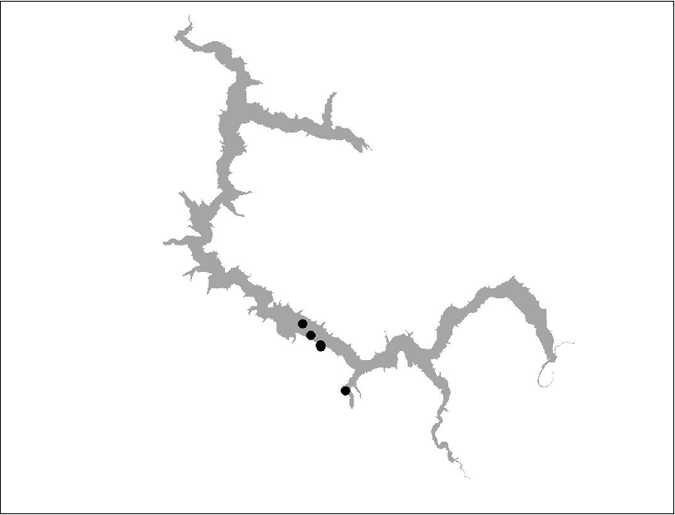
Figure 13 BT 12 (insufficient data)
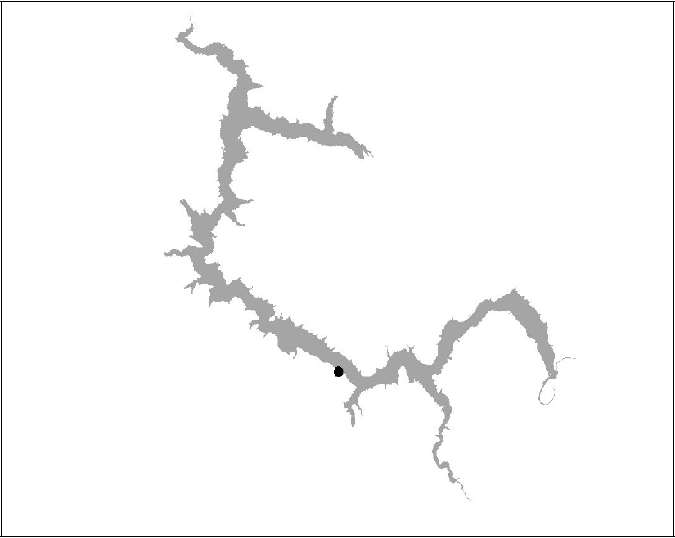
Figure 14 BT15 insufficient data (Died)
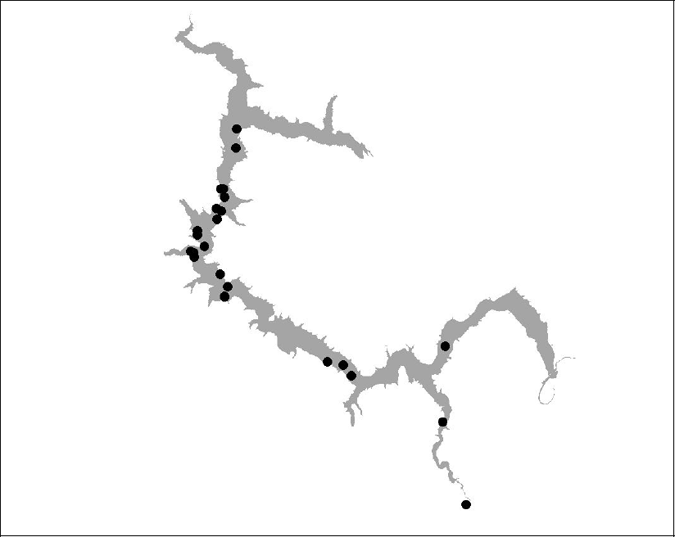
Figure 15 BT 16
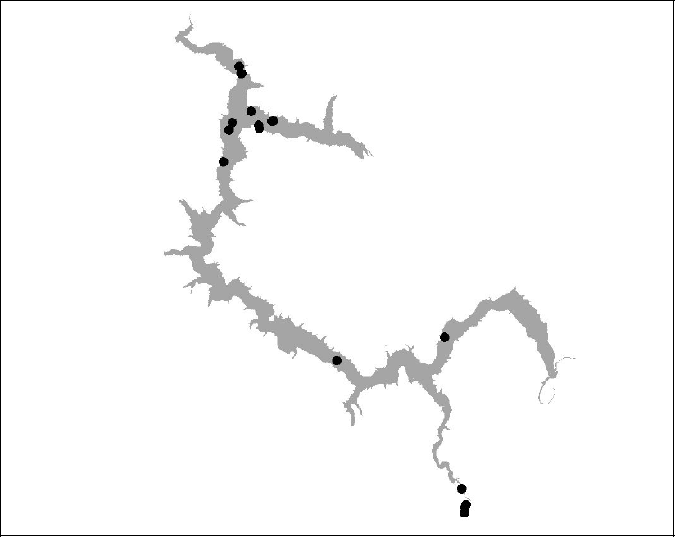
Figure 16 BT 17
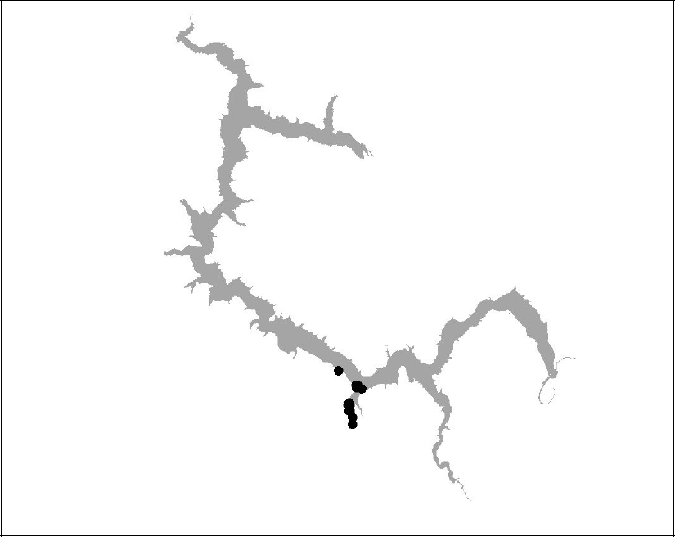
Figure 17 BT 18 (Insufficient data-died)
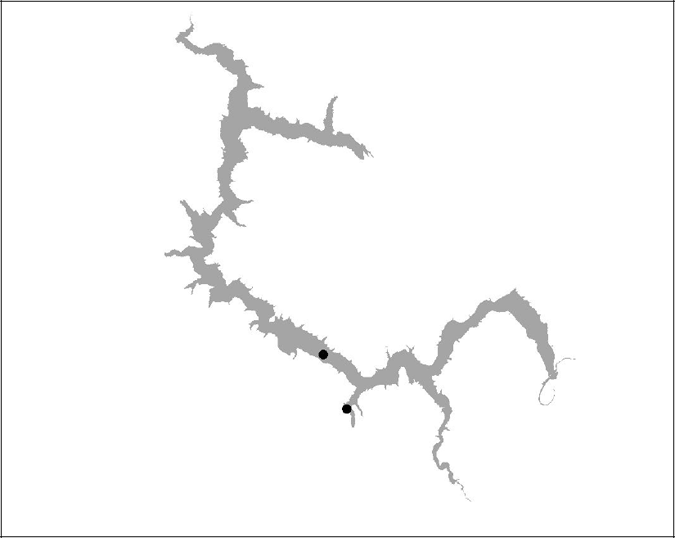
Figure 18 BBT 19 (insufficient data)
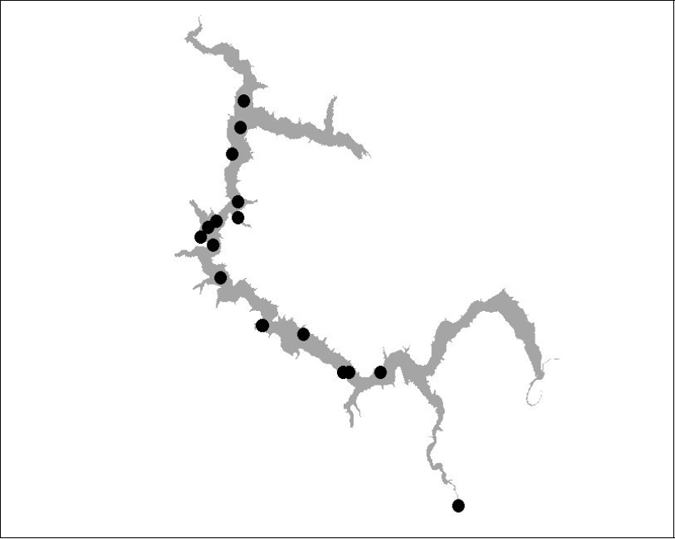
Figure 19 BT 20
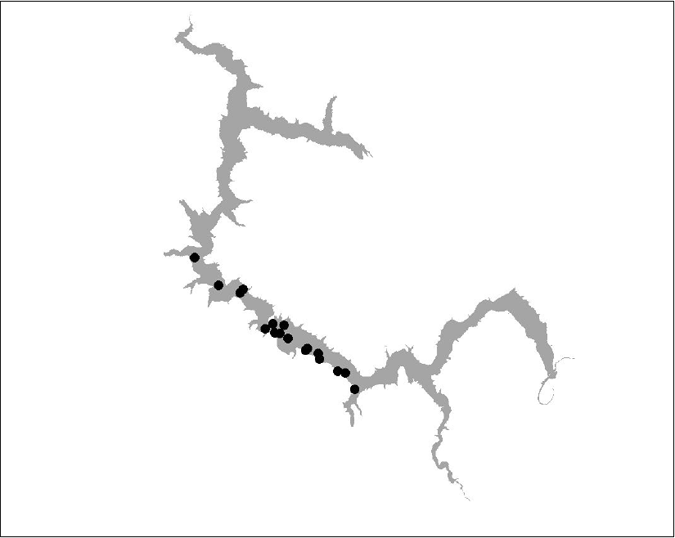
Figure 20 BT 21
Appendix 2
Abstract of paper sent to Canadian Journal of Fisheries and Aquatic Science
Spatial modelling of seasonal changes in brown trout (Salmo trutta L.) habitat in a stratified temperate reservoir in Victoria, Australia.
John Douglas and Nigel W Abery
Abstract
The amount and quality of available habitat can limit fish populations. Large fluctuations in habitat availability could be an important factor influencing fish distribution, population and production levels in temperate lakes. The vertical distribution of trout in lake environments is often related to the critical water temperature and oxygen tolerance of the species. Vertical stratification of the water column is common in many temperate lakes, particularly in summer, and can result in a habitat compression as the suitable trout habitat is squeezed between low oxygen in the lower levels and high water temperatures in the surface layer. The impact of stratification on the size and location of suitable brown trout habitat was modelled for a large impoundment. Species specific temperature and oxygen tolerance ranges were applied to the hydrological profile and lake bathymetry to give an insight into how the stratification of lakes influences the amount and distribution of trout habitat. Habitat was reduced in the study lake by just over 70% in summer. The model was validated with position data from acoustically tagged large brown trout. Such knowledge allows assessment of fish habitat on the whole lake spatial scale, as well as possible interactions between fish population attributes and habitat availability. The method could be applied to a range of fish species and has applications in ecosystem monitoring and fisheries management.
